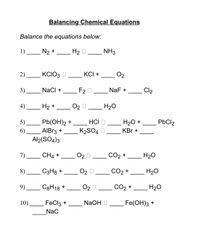
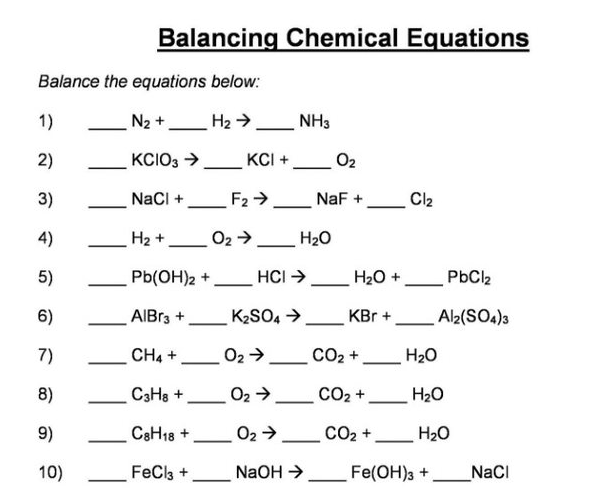
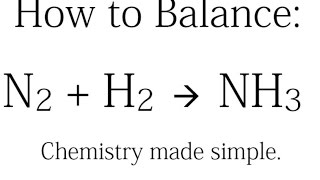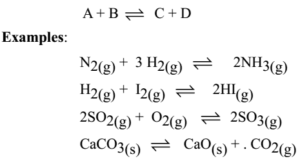N2 +H2 =NH3 Balanced Equation - n2 + h2 → nh3 balance||Nitrogen+Hydrogen=Ammonia Balanced Equation - YouTube
N2 + 3H2 = 2NH3 in above reaction equivalent weight of NH3 and H2 are 1) M÷3,M÷3 2) M÷2,M÷3 3)M÷3,M÷3 4)M÷2,M÷

The chemical equation below shows the formation of ammonia from nitrogen and hydrogen gases. N2 + H2 - Brainly.com

Oxidation Number method. N2+H2=NH3. Balance the chemical equation by oxidation Number method. - YouTube

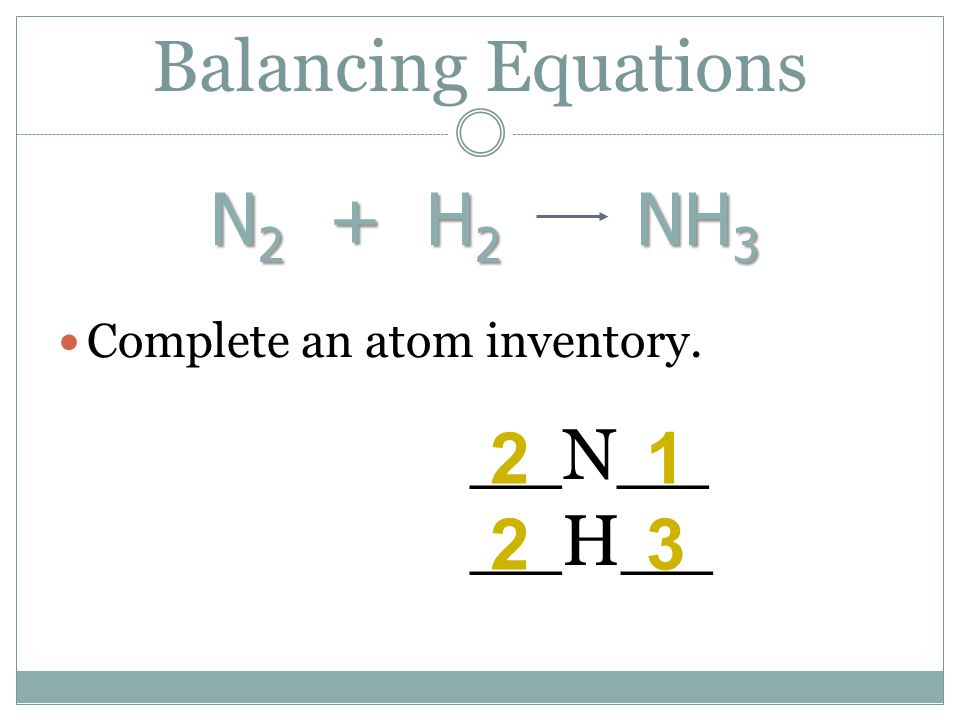
How to balance N2 + H2 →NH3, Nitrogen + Hydrogen = Ammonia balance|N2+H2=NH3 balanced equation - YouTube

SOLVED: The equation 1/2 N2 + 3/2 H2 —> NH3 is not appropriately written because the number of atoms of each element on each side is not the same. fractional coefficients are

N2+H2=NH3 balance the chemical equation @mydocumentary838. n2+h2=nh3 balance the chemical equation. - YouTube

How to balance N2 + H2 →NH3, Nitrogen + Hydrogen = Ammonia balance|N2+H2=NH3 balanced equation - YouTube
![SOLVED: N2(g) + H2(g) <=====> NH3(g) + 94 kJ The equilibrium law expression for the balanced chemical equation would be Question 5 options: [NH3]2 / [H2 ]3[N2] [N2][H2] / [NH3] [NH3] / [H2][N2] [ SOLVED: N2(g) + H2(g) <=====> NH3(g) + 94 kJ The equilibrium law expression for the balanced chemical equation would be Question 5 options: [NH3]2 / [H2 ]3[N2] [N2][H2] / [NH3] [NH3] / [H2][N2] [](https://cdn.numerade.com/ask_previews/ca2e36e2-9489-476e-877f-1c296dc0e2cc_large.jpg)