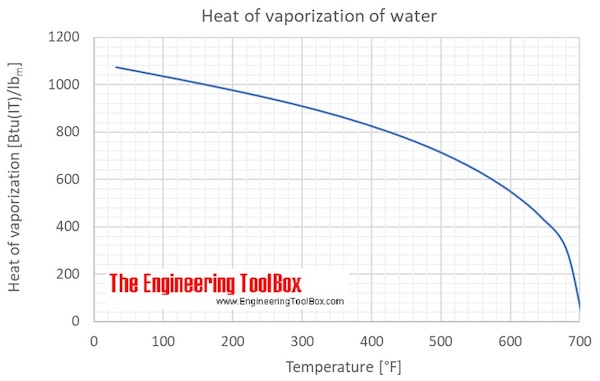
Using Heat of Fusion or Vaporization to Find the Heat Needed to Melt or Boil a Substance | Chemistry | Study.com

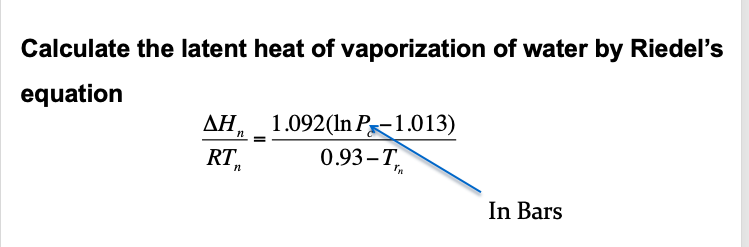
SOLVED: The heat of vaporization of. liquid may be obtained from the following equation: p; In p, TT AH = -R R In Pz T -T p (This is an approximated integrated

The molar enthalpy of vaporization of benzene at its boiling point `(353 K)` is `29.7 KJ//"mole - YouTube